 
The electrons then give off the energy in the form of a piece of light-called a photon-that they had absorbed, to fall back to a lower energy level. If the electrons are given energy (through heat, electricity, light, etc.) the electrons in an atom could absorb energy by jumping to a higher energy level, or excited state. It was identified as a new element in 1781 and first isolated as a metal in 1783. The word tungsten comes from the Swedish language tung sten, which directly translates to heavy. also known as wolfram, is a chemical element with symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively as compounds with other elements. Tungsten is a chemical element of the periodic table with chemical symbol W and atomic number 74 with an. 1 El model de Bohr considera que làtom està format per un nucli atòmic molt petit que conté tota la càrrega positiva i gairebé tota la massa de làtom. While the model had its limitations, it paved the way for further. Tungsten (also called wolfram) 10 11 is a chemical element it has symbol W and atomic number 74. El model atòmic de Bohr és una teoria sobre la constitució dels àtoms, cal citació proposada pel físic danès Niels Bohr lany 1913. It successfully explained the emission spectra of hydrogen and hydrogen-like atoms and introduced the concept of quantized energy levels. The electrons typically have the lowest energy possible, called the ground state. Bohr’s atomic model made significant contributions to our understanding of atomic structure and the behavior of electrons within atoms. The ground state electron configuration of ground state gaseous neutral tungsten is Xe. Unfortunately, there was a serious flaw in the planetary model. 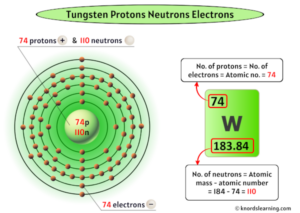
3: Niels Bohr with Albert Einstein at Paul Ehrenfests home in Leiden (December 1925). He suggested that an atomic spectrum is made by the electrons in an atom moving energy levels. Tungsten atoms have 74 electrons and the shell structure is 2.8.18.32.12.2. The solar system or planetary model of the atom was attractive to scientists because it was similar to something with which they were already familiar, namely the solar system. The evidence used to support Bohr's model came from the atomic spectra. \): In Bohr's Model of the atom, electrons absorb energy to move to a higher level and release energy to move to lower levels. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
